How to manage these critical and often misunderstood nutrients
There are some topics that we never seem to tire of investigating, celebrating, or arguing over. You know, in all of humanity, from fertility goddesses painted on cave walls to the newest Cardi B song, there is always more to say about love. While (perhaps) not as arousing, soil has captivated many of us for the past ten thousand years of agriculture. It has been 15 years now that I’ve been in agriculture and working with soils, and yet I continue to enjoy learning more about fertility. What exactly is it that makes the crops rejoice?!
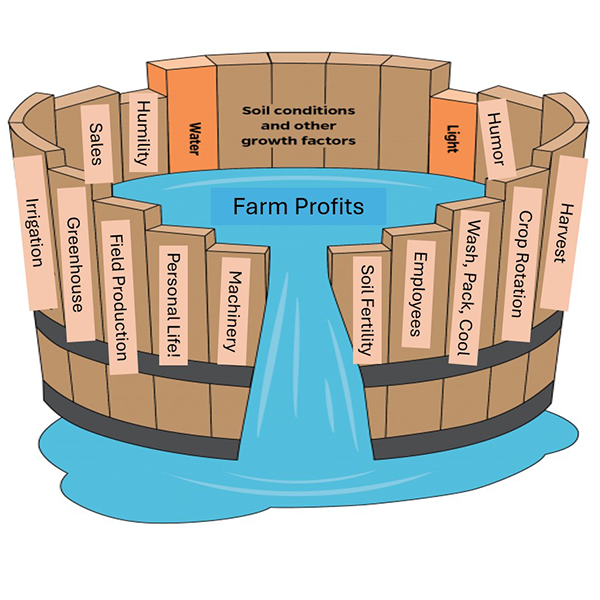
Figure 1: Sam’s Barrel o’ Management, with deference to J. von Liebig and Chris Blanchard. As farmers, we don’t have to be great at too many things, but we do need to be good enough at many different things – just good enough to keep the farm profits (and our personal relationships) thriving. All images courtesy of the author.
We have the opportunity in this article to discuss micronutrients, also called trace minerals, or micros. We’ll talk about where micronutrients fit into the bigger picture of soil fertility, what they are, and how to use them. We’ll address questions like: what are common trace mineral issues, how can you identify them from a soil test, and how to choose and use amendments to improve those issues. Care to join me?
But first, an author’s note: In my experience, many of those who get into market farming and organic agriculture are independent thinkers who want to do things for themselves and who trust themselves. Often this means going against the grain of conventional agriculture and agricultural science. I was such a farmer, focused more on feeding my plants the best rock dust or compost, rather than being sure they had sufficient Nitrogen.
I eschewed much conventional agricultural science as part of the system that I was trying to change. And there was truth to that, but as I’ve grown in experience I have come to understand and appreciate a lot about the conventional soil model. AND — the conventional soil understanding has turned its focus towards biological systems.
From plumbing to machinery to greenhouse growing, there are so many skills to be good-enough-at as a farmer, and it’s a rare person who can perform each of the trades required in farming at a high level (Figure 1). I remember Chris Blanchard talking to us about the ‘management cost’ of decisions, and intensities of management — sure, your greenhouse plants might be slightly better if you fertigated them, but doing so requires another layer of equipment, knowledge, and management. It can be a big win to have systems that are good enough, and require less management.
Consider the management cost
For this article, let’s take a good look at micronutrients — how they work, and how farmers can manage them — but not in a perfect way, just in a good enough way to grow healthy plants and get on with your life, so you can get to packing the delivery truck. I want you to know enough about micronutrients to make your crops thrive, but also enough to know when it’s good enough — and to set your focus on other farm tasks.
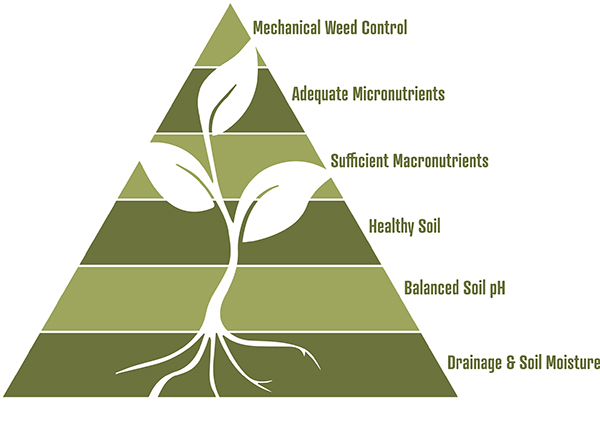
Figure 2: In the plant health pyramid, you can see that to grow healthy crops proper soil drainage and soil moisture is the foundation, then proper soil pH. Once the foundation for healthy crops is set – then attend to soil fertility. Some farmers get excited about trace minerals, before they have attended to more basic agronomic matters. Plants grown in water-logged soil with the wrong pH will not thrive, regardless of the macro or micro nutrients that are applied. From: Weed Management and Cultivation Handbook, Cropline Equipment, 2024.
Let’s start by orienting ourselves with how micros fit into the big picture of soil fertility. I like the plant health pyramid (Figure 2) because it shows the relationship of soil characteristics and their importance. Soil moisture (drainage) is the foundation of the pyramid because it has the greatest impact on soil and plant health and also an outsize impact on the other parts of the pyramid. Whereas micronutrients are towards the top of the pyramid because in a practical sense they are most often less of a limiting factor in production. If you are reading this article: check yourself before you get excited about trace minerals, and confirm first that your soils already have good enough drainage and pH.
What are macronutrients?
Science tells us that plants require around 17 elements for growth. These elements are divided into macronutrients (those that plants require more of), and micronutrients (those that plants require in smaller amounts). In Figure 3 you can see the three macronutrients that plants use the most of are Nitrogen, Phosphorus, and Potassium. These are called primary macronutrients, and most often they are the nutrients limiting plant health.
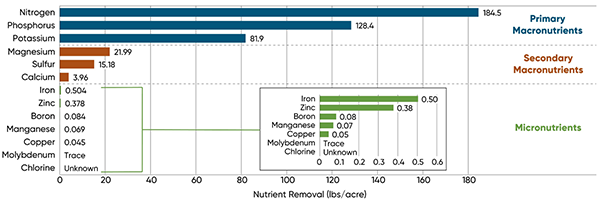
Figure 3: You can see each plant essential nutrient listed in order of the amount in which the plant requires them. Trace minerals really are vanishingly trace amounts – for example, notice that a 300 bushel crop of corn used 2,635 times more Nitrogen than Manganese! (184.5 pounds N per acre / 0.07 pounds Manganese per acre). Heckman et al., 2003. Nutrient removal by corn grain harvest. Agronomy J. 95:587-591.
This is why you hear ag people talk about “N-P-K” so much. Secondary macronutrients are the next three elements most preferred by plants: Magnesium, Sulfur, and Calcium. Then finally we get to the micronutrients, those elements that are still absolutely crucial for plant growth, but are just needed in such tiny amounts.
In fact, plants need such tiny amounts of trace minerals that science did not realize some of them were necessary for plant growth until recently – it happened that scientists assumed the growing media for the experiments was completely devoid of nutrients, but actually trace minerals were present, just in such tiny amounts that they were undetectable at the time – but those tiny amounts were enough to feed the plant.
The micronutrients most often soil tested for are Copper, Iron, Manganese, Zinc, Boron, Chlorine, and Molybdenum. Hopefully Figure 3 shows you why micronutrients are not in the foundation of the plant health pyramid in Figure 2; they are only needed in tiny amounts, whereas plants require much more N, P, and K.
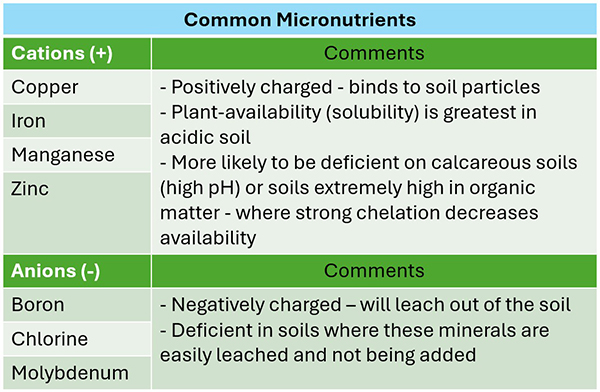
Figure 4: Table showing the charges of common plant-essential micronutrients, and how their charge influences how the mineral moves in the soil, and how it is accessible to plants. Adapted from: Micronutrients for Crop Production, Pioneer Agronomy Services, 2022.
Each of these trace minerals has an electrical charge, which gives each one a unique personality in the soil and plant. These mineral traits will influence what you see in the field and on a soil test. For example negatively charged (-) micros like boron are often the ones to be low in a soil test, since they move out of the soil with water. But (+) micros like manganese may appear low in a soil test while they are actually present in sufficient amounts in the soil. This is because the (-) soil and (+) mineral are bound tightly in a rapturous embrace. Such a tightly bound mineral is said to be chelated, Greek for claw!
The extracting chemicals used in the soil test are not strong enough to break them up, and instead this close affection between manganese and soil endures until soil biology applies its strong grasp on the manganese to feed on it. Such micros are said to be made bioavailable. Though they may appear low in a soil test, they are available out in the field to the plant because soil biology is making them available.
How trace minerals move in the soil
Just like we are attracted to certain people, and thrive when we are around them (looking at you, dear wife), minerals in the soil similarly have certain proclivities towards or away from soil particles. Each mineral in the soil has a charge (+ or -) that gives it a personality in how it moves in the soil. Nutrients that stick around in the soil tend to be more available for plants. Other nutrients tend to be carried away from the soil in water, called leaching. When rain water runs into, through, and out of the soil, it will take certain minerals away with it.
Minerals cycle from the soil to the bodies of plants and animals and back again. While minerals are ‘dead’ in the soil, they exist as either mineral particles from stone, or particles that were formed by living beings, called organic matter. Both mineral and organic soil particles are negatively (-) charged. Many minerals have a positive (+) charge. Because opposites attract, these (+) soil particles will hold tightly to the (-) soil.
This means that (+) minerals like iron are often held in the soil and available for plants. But some plant essential minerals are negatively (-) charged and are repelled from (-) soil particles. These minerals are more likely to get washed away in water, and are commonly less available for plants to access.
Know your region’s soil
Ours is a BIG country, and each region will have unique soil issues based on soil and climate. So while a book or this article can give an overview, an agronomist, consultant, soil lab, or extension specialist in your region will have an actual ‘boots on the ground’ understanding of common nutrient deficiencies. In fact, most states have done lots of research to learn which micronutrients tend to be needed on the native soils and which are rarely deficient. There are also several published recommendations for the amount of each micronutrient needed for good plant growth, such as the ones in Figure 5.

Figure 5: Adapted from T.K. Hartz, UC Davis, 2007.
How to interpret micronutrients from a soil test
Most trace mineral amendments are exactly that — nutrients in the mineral form — they are just different ground-up rocks. And the more finely they are ground, the more quickly they turn into a form that plants can access, but that change is a process that takes time, weather, and soil biology. Science commonly understands measures of micronutrients with the unit of parts per million, or ppm. Ppm means that as an average, out of a million plant-available particles in the soil, that number (x ppm) of them are the mineral which was being tested.
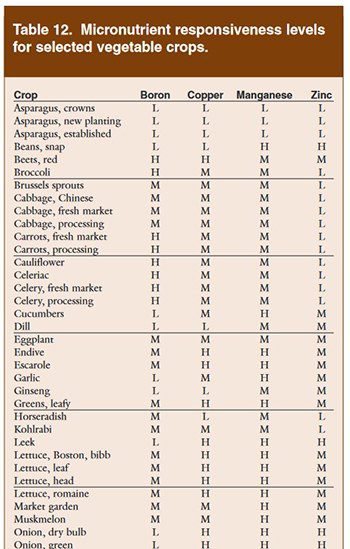
Figure 6: From Nutrient Recommendations for Vegetable Crops in Michigan, MSU, 2004.
My recommendation (and that of a whole lot of experienced people), is to usually add micronutrients to your soil in response to a soil test. Or as John Kempf likes to say, “We don’t guess, we test.” I say that trace minerals should ‘usually’ be added in response to a soil test, because the exception is that some farmers, at the beginning of their soil stewardship, will apply a broad array of trace minerals using rock dusts to make sure a wide spectrum of minerals are present (though as we shall see, in tiny amounts).
While it’s not perfect, a soil test uses chemicals to mimic the root environment, and to guess at which minerals the roots have access to. But that can be hard, remember that minerals are bound more less tightly in the soil: from ones like boron (-) that leach away with water, to iron (+) that are bound (chelated) very tightly to the soil, and everything in between.
When you do a soil test, Figure 7 shows what it might look like. In this soil test from a Wisconsin vegetable farm, you can see the micronutrients in the red box.
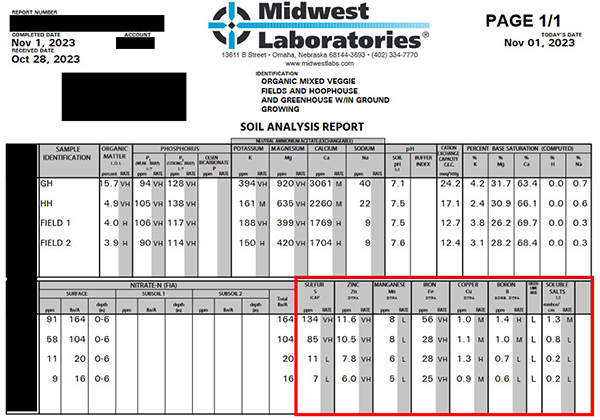
Figure 7: In this soil test from a Wisconsin vegetable farm, you can see the micronutrients in the red box.
I remade the micronutrient table, figure 8, in better focus – let’s look at boron. Each row, from 1.4 ppm down to 0.6 ppm, is from a different field or a hoophouse.
This farm has a range of boron levels in different soils, which is reflected in the ratings. With this information you can combine science with local soil knowledge to choose an amendment and determine how much is needed to fatten your plants. You can ask your soil lab to give you their recommendations for products/amendments and rate of application, you can work with a consultant/agronomist, or you can figure it out yourself. Whoever does it, they will likely look through a resource like the Nutrient Recommendations for Vegetable Crops in Michigan (from Figure 6), and use the information for their recommendation. If I was working with this farmer on their soil test and thinking about boron, I’d summarize the important points about boron from the MSU guide like this:
Boron recommendations are based on crop observations, not on soil tests, because in the soil boron is water-soluble and it leaches out of the soil with the movement of water. As a result, the plant-available boron changes throughout the season with soil moisture, especially in sandy soils. For crops that often show improvement from added boron, such as broccoli, cauliflower, celery, beets, turnips and rutabagas, boron deficiency may occur when soil moisture gets low. Even though the soil contains enough boron, it’s just not available to the plant. Applying 2 pounds of boron per acre per year is recommended for these responsive crops grown on sandy soils (CEC <8.0). Boron may be applied in the broadcast fertilizer or included in the starter fertilizer. Applying boron to the foliage can be effective. Apply 0.3 pounds of boron per acre. Spraying more than 0.5 pounds of boron per acre in one application may cause foliar burn. Never apply boron, soil or foliar, to sensitive crops such as beans, cucumbers and peas.
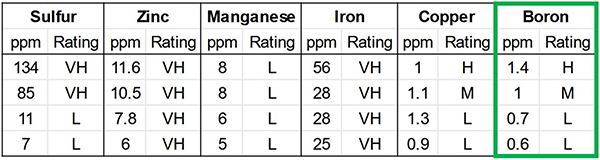
Figure 8: I remade the micronutrient table in better focus – let’s look at boron. Each row, from 1.4 ppm down to 0.6 ppm, is from a different field or a hoophouse, Low (L), Medium (M), High (H) or Very High (VH). This is the micronutrient table from the soil test in Figure 7.
We can see on the soil test that this farm’s boron levels range from low (L) to High (H), and we can decide how much boron we need to buy, which fields need it, and how much we would benefit from applying. And, knowing the rate we need can also help us determine which amendment to use.
Using micronutrient amendments
For any amendment that you will buy from a reputable company, you should know exactly what is in it. The producers of quality amendments publish an analysis, which is the results of a third-party lab showing exactly what minerals/chemicals/nutrients are present in their amendment and in what amounts. An analysis is often given in percent by weight. As in, “My 100 pound bag of product contains 10% ____(mineral) by weight.” Since 10 percent of 100 pounds is 10 pounds, we know the bag contains 10 pounds of that mineral. But because trace minerals are needed in such small amounts, they are often shown in an analysis by ppm (parts per million).
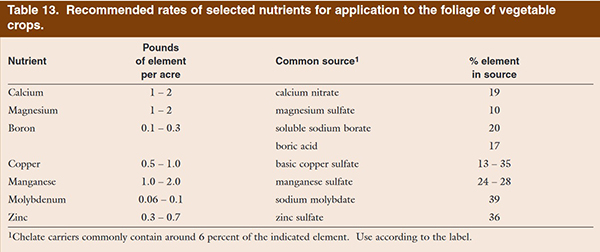
Figure 9: A table that I might rely on when helping a farmer with a trace mineral fertilizing program. From Nutrient Recommendations for Vegetable Crops in Michigan, MSU, 2004.
Let’s look at a popular trace mineral amendment: Azomite. I used to use it when I was a young farmer. Azomite is a mined rock left over from an ancient volcano in the western United States. It contains many minerals. The description of many companies says: “Contains trace minerals and elements including Boron, Calcium, Chlorine, Cobalt, Copper, Iron, Magnesium, Molybdenum, Nitrogen, Phosphorous, Potassium, Silicon, Sodium, Sulfur, and Zinc.” And it is true that Azomite “contains” those micros, but how much?!
If we are concerned with adding boron at a rate of 2 pounds boron per acre, thanks to the internet I can look up the analysis of Azomite and find that it has around 29 ppm of boron. That’s uh, a trace amount! If I apply Azomite at their recommended rate for vegetables, 150 pounds per acre, and that batch contains boron at 29 ppm, then the 150 pounds of Azomite has about 0.004 pounds of boron. But I need 2 pounds of boron!
In fact, you would need to apply around 68,000 pounds of Azomite per acre to meet the recommendation of 2 pounds of boron per acre. So sure, Azomite “contains” boron, but it does NOT contain ENOUGH. Hopefully this illustrates how you (or your soil consultant) can think through choosing micronutrient amendments. Azomite is a quality product and contains many other nutrients at varying levels — for another soil or for a different mineral, this amendment might improve plant health.
Next let’s look at Sea-90, this amendment is made from evaporating sea water: it is sea salt. Similar to mined salt, but just several million years more recently suspended in sea water. Because Sea-90 is a quality product, we can look up the analysis for it and see what minerals it has and in what amounts. With boron as our continued example, Sea-90 has around 20 ppm boron. If we think back to our Azomite calculations, to apply our recommendation of 2 pounds of boron per acre, using Sea-90 to do it would require around 100,000 pounds of Sea-90.
Let’s look at one final amendment. Called Solubor, it is available as a powder that dissolves in water. It is 20 percent boron. So a 50 pound bag of Solubor has 10 pounds of boron in it. So 10 pounds of Solubor would supply the 2 pounds of boron that I want. If I was helping a farmer select a trace mineral amendment to increase the soil’s boron, I would be more likely to recommend a product like Solubor, before Azomite or Sea-90. Whereas those products could work well on other farms or for other minerals.
Using trace mineral amendments
There are two main ways that farmers apply trace minerals:
1. A homogenous trace mineral package that goes out on all fields every year. It is a single mix of several amendments, and includes minerals that are often low in your farm’s soils, like boron. These can be custom blended into your fertilizer, and applied by broadcasting or as a seed treatment.
2. Targeting specific fields or crops that tend to be low in specific micros.
a. Amendments are used to apply only the micros needed, at the amount they are needed.
b. These can be applied by broadcasting or as a seed treatment.
c. Often this will include foliar spraying, because foliar spraying is the best way to apply many micros.
What to do when you put this article down
If you want to take another look at your micronutrients after reading this article, you could:
- Take accurate soil samples
- Get soil tests from a lab that knows your region’s soils
- Use that soil test to choose trace minerals and their rates by speaking to a consultant, agronomist, extension specialist, etc.
- Or consult your state’s references to figure out your own micronutrient recommendations
I have learned from many great resources and kind and experienced ag professionals who took the time to speak with me for this article. Thank you to Alan Philo, an agronomist with years of experience making nutrient recommendations to Midwest organic farmers. Alan wrote a really neat article on how soil works, find a link below. Dan Fillius of Iowa State Extension is a gem of a specialist, and shares what he learns from visiting so many vegetable farmers. Read it here: tinyurl.com/mwkhxs3h. Erica and Alice from Grow Abundant Gardens shared their passion and scholarship for plant nutrition.
Sam Oschwald Tilton is an organic agronomist, and helps farmers improve their weed management, soil, and machinery systems. He also delivers hands-on trainings and works with organizations to develop resources, through his business Glacial Drift Enterprises. He also organizes the Midwest Mechanical Weed Control Field Day (August 20th this year in Ames, IA). Sam lives in Minneapolis and enjoys gardening with the neighborhood. If you want to talk to Sam please email him – glacialdrift@protonmail.com.
Copyright Growing For Market Magazine.
All rights reserved. No portion of this article may be copied
in any manner for use other than by the subscriber without
permission from the publisher.











