What it is, why you care and how to manage it
Soil pH can have a huge effect on your plant health, but because it’s not always a problem it is often one of the lesser things to concern a farmer. Like many of the aspects involved in making a living growing direct-market crops, if you can get by without knowing too much about soil pH then don’t spend too much time on it.
With the current state of soil science and soil testing knowledge, you can send in a soil test every four-ish years, pay about $30 per field/plot, and get an accurate recommendation of a rate to adjust your soil’s pH. However, if you’ve got the time, I’ve got a story for you. In the course of this article we will appreciate pH more fully for the beauty and mystery it reflects, and I’ll share what I think about when I am advising a farmer.
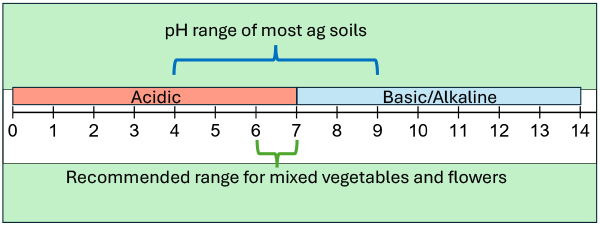
Figure 1: Important parts of the pH scale, by Sam OT. A note about the pH scale is that it is logarithmic, this means that between pH 6 to 7, there is a 10x difference in the concentration of H+, or between pH 5 to 7, there is a 100x difference. You can see why it can be a big deal if your soil was at pH 5.5 instead of a target of pH 6.5.
I’ll weave together a story showing: what pH is, why you should care, and how you can manage it. You’ll see the neat connections between chemistry and hands-on agriculture. As an enticement, let me share the words of esteemed University of Wisconsin-Madison soil science professor Emil Truog: “If you could have but one soil test run on a field, that test should be pH.”
But know that if I succeed in telling a compelling tale, it is because there are facets of pH that I have elided or simplified, and that continue to confuse and intrigue soil scientists. I should also say that soils around the world are wonderfully diverse, and my knowledge of soils reflects my midwestern ag experience. Specifically, soils in tropical areas may not behave in the ways I am describing.
What is soil pH?
There are so many lenses through which to understand how nutrients behave in soil. While the biological aspect is crucial (and was neglected by mainstream agriculture from maybe 1945 to 2005), to understand soil pH we’ll look at the plant and soil through a more chemical lens.
You know how there are substances/chemicals like CO2 (carbon dioxide) or H2O (water), and then there are individual elements that make up those chemicals, for example H+ is free hydrogen. These chemicals and elements all have charges: positive or negative (think magnets or batteries). And, they also have different strengths of attraction — each is more or less positive/negative than others. For example, H+ has a higher strength of attraction to negatively charged particles than does K+ (potassium). These charged nutrients are called ions, and some of them are plant food, mineral nutrients.
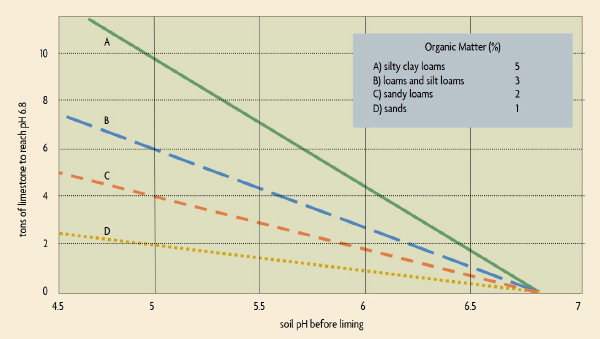
Figure 2: You can see in this graph that clay and silty soils are more resistant to changing pH than sandy soils. Their reserve acidity is higher, they are more buffered, and they require more limestone to get the pH to a given level. Adapted from the great book Building Soils for Better Crops by Fred Magdoff and Harold van Es.
These mineral nutrients exist in the soil in about three different places: On soil particles, dissolved in soil water (also called the soil solution), and in the bodies of microbes and plants (to be released when they die).
Please allow me to paint a picture of the soil solution, so that you understand why I think it is fire. An aqueous film surrounds all the living things and the individual particles that make up a soil. It’s like at the end of doing dishes you drain the sink and there’s a final fork at the bottom, it’s covered in a grimy film of water into which are dissolved all manner of organic substances. That fork, with a film of gooey water around it, is a soil particle.
Only in extreme drought does the soil dry out enough to remove this watery film, within which is dissolved all manner of: living microbes, uncountable biological substances exuded from plants and soil-life, and mineral nutrients. When these mineral nutrients are not dissolved in the soil solution, they are in various forms of exchange among: soil particles, living microbes absorbing them, dead microbes releasing them, roots absorbing them, and roots releasing them. Mmmm, the soil solution — a frothy brew indeed.
Because mineral nutrients are constantly shifting between these three places (soil particles, the soil solution, and the bodies of microbes and plants), the amounts available to plants are never static. The amounts of mineral nutrients available to plants are always changing with factors like temperature, oxygen, water, biological activity, and pH.
It’s like we are dealing with a rushing river that is always changing, but to tell this story in a few pages with clarity, I am taking a photo of this river, freezing its surging multitudes in time and space, and telling you that the photo truly represents the river. Ah, the grandeur of creation and the limitations of the human mind.
Okay, turning from the soil solution and whether we can really know it, back to the charges of the mineral nutrients, like H+ (hydrogen). Being aware of the charges of mineral nutrients is a neat way to understand how each behaves in the soil, and how they’re made available to plants — their personalities, if I may.
All plant nutrients share a character trait with your yoga instructor because the soil demands chemical balance. The electrical charges in the soil have to balance. This means that if a plant root wants to absorb a delicious negatively-charged nitrate ion (NO3-), it needs to give off enough positive ions into the soil solution to equal the negative charge of that sweet nitrate that it is absorbing.
Plants, those wonders of nature, move a lot of nutrients around using free hydrogen (H+). Plants have a lot of H+ that they don’t need and can use in exchange. And this H+ is what roots are often pumping into the soil to balance out the negatively charged particles that they find so delicious. But plant exudates are not the only process adding H+ to the soil. Rain, the soil atmosphere, soil biology, the leaching of water through the soil, and many synthetic fertilizers all have the effect of adding H+ to the soil, where it can accumulate on soil particles or dissolve into the soil solution.
That H+ hydrogen ion is sort of a default ion in the soil; it fills in wherever needed to balance charges. And, it is happy to take the place of other positively charged ions because it has a higher strength of attraction to negative particles than most other positive ions. For example, in a lot of cases, due to their strength of attraction, positive ions will bind in this order to negative particles: first hydrogen (H+), then calcium (Ca2+), then magnesium (Mg2+), then ammonium (NH4+).
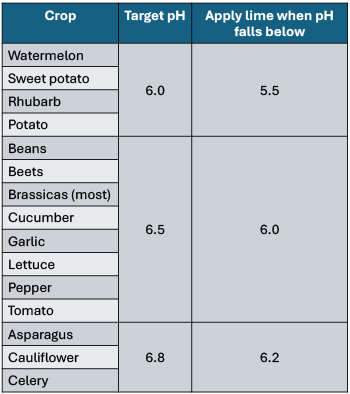
Figure 3: Target pH ranges for some crops in mineral soils based on field research under conventional management. By Sam OT, adapted from: Nutrient Recommendations for Vegetable Crops in Michigan and Mid-Atlantic Commercial Vegetable Production Recommendations.
Why do you care? Because this is the order that nutrients will move out of your soil. Because it has a strong strength of attraction, you’ll notice that hydrogen is one of the last positive ions to move out of your soil. It can be the “last man standing” when other ions have left.
What would happen, heaven forbid, if H+ took the place of most of your positively-charged mineral nutrients on soil particles? Then, you’d have fewer nutrients available for your plants, and even if you added them to your soil as fertilizers, they wouldn’t stick around because they’d have less space on the soil particles to stick onto. Or what if there was too much H+ in your soil solution? Then, the microbes that should be vivaciously living their best lives and making nutrients available for your plants would be lethargic or dead because they’d be living in an acidic soil solution.
Why you should care
Now, we have arrived at the chemical heart of pH from a plant-centric perspective. pH is the “potential Hydrogen” how much H+ is floating around the soil solution. The presence of H+ in the soil solution can have a big impact on the microbes that live there, and their functioning biologically impacts the nutrients available to your plants.
At the same time, when the concentration of H+ gets too low (acidic) or too high (alkaline/basic) it can chemically change the availability of mineral nutrients to your plants, resulting in deficiencies or toxicities. Free hydrogen plays such a big role in whether mineral nutrients are available to plants that we invented the whole pH scale to measure it.
Your soil’s pH directly affects: microbial activity, the availability of most plant-essential nutrients, the soil’s ability to hold positively-charged nutrients, the appearance of plant-toxic heavy metals, and the performance of some herbicides. Boom, now are you glad you read this far?!
When you receive your soil’s pH from a soil report, it is measuring the pH of the soil solution, called the active acidity. Remember my analogy of the river, and limits of our human understanding? Looking at active acidity alone is like taking a picture of the river, or like knowing the temperature outside at 8:53 a.m. It’s helpful, but it’s not the full picture.
While active acidity tells us about the amount of hydrogen (H+) dissolved in soil water, it does not tell us about how much H+ is stuck onto soil particles. The amount of H+ held on soil particles is called reserve acidity. And it’s important because if you have lots of hydrogen held on soil particles (high reserve acidity) but you’re only paying attention to the H+ in your soil solution (active acidity), then when you try to change the pH there will be lots of “reserve” H+ in soil particles that will flood the soil solution and keep your soil pH right where it had been.
Reserve acidity makes soils buffered — resistant to change. Reserve acidity is largely dependent on: 1. the amount of organic matter in your soil, 2. the soil texture (sand, silt, clay), and 3. the amount of hydrogen held on soil particles. Soils with higher organic matter or clay tend to be more resistant to changing pH than sandy soils. If you want an accurate pH recommendation, you need to have them test not only “soil pH”, but also “buffer pH.” Then, your recommendation will take into account both the soil’s active and reserve acidity.
How you can manage pH
Bacteria are generally happier at higher pH and fungi at lower pH. I’ve talked to experienced agronomists who observe the most biological activity around pH 6.8. Science says that a good pH range for most soil organisms is between pH 6.0 to 7. Because a soil’s biology plays a huge role in making nutrients available to plants, keeping microbes and fungi happy keeps your plants happy.
Subsequently, pH 6 to 7 is generally a fine range for mixed vegetables and flowers. If you’d like a tighter range consider pH 6.8, as this is soil biology’s happy place. While you can dial-in to a more specific range than pH 6 to 7, for mixed crops it may be unnecessary and cause unneeded expense.
However, if you grow just a few crops in a clearly defined rotation, it may benefit you to adjust your soil to a more precise pH based on your crop’s preferences. Each vegetable species has a range of soil pH where it thrives, based on the soil pH of its motherland. For farmers growing a few crops, a good strategy to keep things simple is to choose a target pH for your fields based on your most acid-sensitive crop (crop with the highest target pH).
For example, looking at Figure 3, if I were growing primarily beans, cauliflower, and watermelon, I could choose a target pH of around 6.8, because cauliflower is my most acid-sensitive crop.
You can also sometimes use soil pH to manage plant disease because the microbes that cause certain plant diseases do not thrive outside of their preferred pH range. Potato scab, for example, is controlled by maintaining soil pH at 5.3 or below, whereas club root disease of cabbage and cauliflower can be controlled with a pH of 7.2 or above.
Let’s assume you grow a mixture of many vegetable or flower species with multiple crops per season on each plot, and, you trust ol’ Sam and choose to manage your soil between pH 6 to 7. You send in your soil samples, ask for a “buffer pH” analysis, and get a recommendation back from your lab for a material and rate to get your pH within that desired range. If your soil’s pH is getting low (acidic), then the lab will often recommend some type of limestone to adjust it. Let’s talk about a few things you’ll want to consider when adjusting your soil’s pH:
Sampling depth: Your soil lab will give you instructions on how deep to take soil samples. Following this is crucial, because all of the lab’s recommendations are calculated for this volume of soil. A depth of 9 inches is typical; it’s based on the average depth farmers used to plow. The lab is assuming that you want to change the pH in this 9-inch deep volume of soil, and that you will incorporate the lime to that depth.
If you want to change the soil pH deeper than 9-inches deep or less than 9 inches, then another calculation should be done so that the liming recommendation reflects your actual tillage system. Clemson extension has a nice calculator to adjust lime rate for tillage depth at https://rb.gy/hqq6bp.
Do your plants only root to 9-inches deep? I hope not; I hope they have access to the vast stores of nutrients and moisture below 9 inches (except the shallow-rooted crops). If you are first getting to know your soil, consider doing two soil tests at different depths. This way you can understand the pH of your topsoil and your subsoil.
If your subsoil’s pH is way off, your plant roots and microbes are discouraged from venturing into those depths. Depending on your results you might work with an agronomist to adjust the pH of both your topsoil and subsoil through liming and possibly deeper tillage.
Tillage system: This relates to the discussion above on sampling depth. In your tillage system, to what depth do you commonly till: 9 inches, 6 inches, ½ inch? If you get a lime recommendation, it is meant to be incorporated to the sampling depth. If you are no-till but use a lime rate based on a 9-inch sampling depth, you will be applying 9-times too much lime to the top 1 inch of your soil.
When lime is applied only on the surface and not incorporated, it can take 10 years for it to change the soil pH at a 6-inch depth. The more you incorporate your lime, the quicker it will work. Also, if no-till growers are leaving all amendments at the surface, their soil-surface pH at 0 to 2 inch depth might be a lot different than the soil below that. Just like some farmers may want to understand their subsoil pH, no-tillers may want to understand their near-surface soil pH. Adjust your lime rate to match your tillage system.
Irrigation water: There are many regions in the country where the ground water sits in limestone aquifers. This means that every time you irrigate you are adding a little lime to your soil. For those who are irrigating often, you may want to account for the lime in your irrigation water when choosing a lime rate.
Choosing a material: I will avoid too much detail here, but I think the most important point is that the finer the limestone is ground, the quicker it will change a soil’s pH. I tend to recommend veggie/flower farmers use finely ground and high-purity lime to adjust their pH more quickly. Because your crops are higher-value than row crops they can justify the greater expense of the best lime material to adjust your pH the quickest.
Safety first! Finely ground lime lingers in the air, please learn from my dear brother Joel, who didn’t have the benefit of this article when making his first lime application without goggles – he lost some vision from the lime dust and was unable to read for several weeks. Thank god, his sight returned and he had the opportunity to learn from that mistake.
When to apply: Unlike some nutrients (looking at you nitrogen), lime is not lost to the soil through leaching. So, you can apply it any time during the season when you have time or it fits with other field operations. You can apply lime with other fertilizers as long as it gets spread evenly. Lime generally begins affecting soil pH in a few months and will have full effect in two to three years.
So, a good place in your rotation to apply lime is after your most acid-sensitive crop, so that it has as long as possible to react and adjust the soil pH before that crop is grown again. Farmers generally don’t want to apply more than two to four tons of lime per acre in a single season. If you need more than that then split it over two seasons.
We did it
Alright, we did it, all in a single article. Now, what exactly we did is for you to judge. I hope to have shared a story of soil pH so that you understand your soil a little better. Also to have given you hands-on pH management tips that you can use. Don’t feel like you have to know it all. Your soil lab, extension agent, or ag advisor can help.
“But Sam!” you say, “While you’ve discussed soil acidity and liming, what about those of us on calcareous soil who have no issue with too much H+, but rather our pH is already too high?!” Ah, you wily reader, I can’t slip one past you. Well, to address soil with a pH that is too high, you’ll have to implore our illustrious publisher to commission another edition of – Soil Story Time with Sam. Until then I remain, yours, Sam OT.
Sam Oschwald Tilton is a long-time veg head, and an organic agronomist. He helps new and established farmers improve their weed management, soil, and machinery. He also delivers hands-on trainings. Sam lives in Minneapolis and enjoys gardening with the neighborhood. You can contact him at glacialdrift@protonmail.com.
Copyright Growing For Market Magazine.
All rights reserved. No portion of this article may be copied
in any manner for use other than by the subscriber without
permission from the publisher.











