Dear plant and soil tenders, in the past few issues of this magazine we looked at managing the soil’s micronutrients and pH. Those topics were micro. Now, our goal for deeper knowledge is macronutrients — those nutrients that plants use the most of. How could one writer possibly share some helpful theory and practical tips about nine plant nutrients, all in one article? One mortal writer is not enough. No, to do macronutrients justice two soil nerds of the first degree teamed up. We will discuss how nutrients move in the soil and the plant, common issues to watch out for, and big-picture questions. Come along, won’t you?
Let’s start by considering soil nutrients in the context of a complex commercial farm. While macronutrients are crucial for healthy plants, they are one of several components of soil management that promote bountiful crops. The illustration below suggests that it won’t serve you to nerd out about micronutrients (or even macronutrients) without also attending to more foundational soil issues: soil drainage/moisture/irrigation, pH, and biological activity.
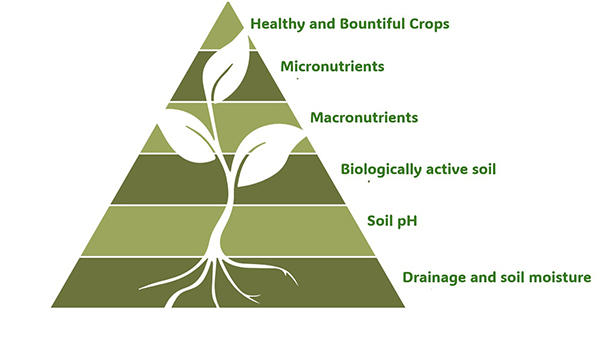
It won’t serve you to nerd out about micronutrients- or even macronutrients- without also attending to more foundational soil issues- soil drainage/moisture/irrigation, pH, and biological activity.
Now that we’ve understood macronutrients as part of the bigger picture of plant health, let’s recognize the diversity of mindsets on soil fertility, because not everyone will agree with how we approach this article (and that’s okay). On one hand, soil fertility can be perceived through a purely chemical lens, where individual nutrients and their function are identified and singled out to make fertility management. It’s like following a recipe, where you find the limiting ingredient and add it. On the other hand, soil fertility is regarded as infinitely complex natural processes that should be managed holistically, focusing on increasing organic matter, providing enough moisture, and inoculating soil with “good biology.”
We have learned from growers, researchers, and advisors with various mindsets on the topic, and try to keep our ear to the ground and stay open to new perspectives and data. That being said, we’ve both worked with farmers who have taken a more vibes-based approach to fertility management for several years, which resulted in real nutrient-related challenges that impact crop quality, pest and disease pressure, and yield. We’ve also seen a vibes-based fertility approach grow great crops. In managing soils with an eye toward preventing and solving these problems, we try to acknowledge both the ecological and chemical ways of understanding soil fertility.
Whatever your mindset, there are some basic fertility rules of thumb that are crucial to understand. Growers can get by with those basics and a handful of extension publications. Farmers don’t have to wrap their minds around the exact science of it all. There are plenty of expert advisors available to help you make an actionable nutrient management plan.
A good advisor can be your educated sounding board, helping you focus on specific problems and find real-world solutions. And unlike a farmer, outside ag advisors are usually not also having to make a seed order, fix the tractor, update the OSP, re-skin the high tunnel, apply for a spot at the new market, hire next season’s crew, and order salad bags by…yesterday.
Dialing-in fertilizer applications
Because mixed vegetable and flower farmers grow so many darn crops, often in several different fields, it’s tempting to have a blanket rate of compost and/or fertilizer to apply everywhere regardless of each field’s soil test results or planned crops. However, your fields might have varying pH values or contain vastly different nutrient levels, and a head of lettuce needs way less nutrients than a tomato plant.
A farm’s production often benefits greatly from basing fertilizer application rates on each field’s soil test result, production history, planned crops, and soil type. Disregarding those details can cause a host of problems that we have seen in both our personal farming experiences and with farms we have worked with.
Macronutrient ‘personalities’
Generally, 90 percent of a plant is made from carbon and oxygen. In a wonderful lightening of the farmer’s load, the air (carbon dioxide, aka CO2) and water (aka H20) are the plant’s sources of carbon, oxygen, and hydrogen (the 3rd structural element). But outside of carbon, oxygen, and hydrogen, fertile soil is the medium responsible for supplying the other six macronutrients to plants.
Nitrogen (N), phosphorus (P), and potassium (K) are the primary macronutrients. They are named as such because plants use more of them than the secondary macronutrients: calcium (Ca), magnesium (Mg), sulfur (S). Soils themselves have their own native mix of macronutrients based on their land-use history, and before that, their geology — the stones and particles from which they are formed.
For example, soils in Kentucky’s Bluegrass region are high in both calcium and phosphorus, because their parent material is a limestone that contains calcium phosphate minerals. But, a native soil that is naturally low in calcium and phosphorus could have off-the-charts levels of both if it had a long history of repeated chicken litter applications. While it’s not necessary to know the intricate details of chemical and biological processes that move soil nutrients, it is helpful to understand the basics: how each macronutrient moves in the soil, is absorbed by plants, and moves within the plant. Knowing these nutrient characteristics and your soil’s macronutrient levels will improve your overall nutrient management.
Nitrogen
Nitrogen (N) is the most common limiting nutrient for plants. An awareness of how plant-available forms of N are converted from and into different forms that move through the air, soil, and plants can help you make the most of your fertilizer, save money, and prevent pollution. Here are the N cycle processes, quick and dirty (how farmers like it):
Fixation: Nitrogen gas (N2) surrounds us in the atmosphere and is not accessible to plants until it is fixed in the soil or converted into other forms of nitrogen. In organic systems, this is most often done by bacteria in the root nodules of legumes, called rhizobium, which convert nitrogen gas to ammonia (NH3) or ammonium (NH4+). Ammonium has a positive charge, so it sticks to clay and organic matter particles in the soil, preventing it from leaching.
Mineralization: N that is bound to the soil’s organic matter (decaying plant and animal tissues, manure) is released by microbes and converted to plant-available forms of N (also called inorganic N). Understanding this allows you to reduce the rates of N fertilizer applied to soils with high organic matter content — saving you money.
Nitrification: Nitrifying bacteria convert ammonia to nitrite (briefly) and then nitrate (NO3-). Nitrate has a negative charge and it’s not attracted to clay and organic matter particles. It hangs out in the soil solution, which is both a pro (more available to plants) and a con (more susceptible to leaching, or draining down and out of the soil with water).
Denitrification: When the soil is saturated with water and there is not enough oxygen for microbes, the microbes use nitrate as their source of oxygen, converting nitrate to nitrogen gas or — yikes — nitrous oxide or nitric oxide. The latter are potent greenhouse gasses. Either way, waterlogged soil makes N go bye-bye.
Immobilization: Soil microbes take up nitrate and ammonium, temporarily making them unavailable to crops. An example is when high-carbon plant residues (like straw) are incorporated into the soil, then microbes “tie-up” the available nitrogen as fuel to break down the carbon. Immobilization is inconvenient in the short term and in some cases undesirable because it makes that N unavailable to crops. However, it can pay longer-term dividends as it provides a slow-release of that N.
Leaching: Nitrate hangs out in the soil solution, dissolved in soil water. In a heavy rainfall, nitrate will move with water into deeper soil below the crop’s roots or into groundwater and streams, rivers, and lakes. In this scenario, the dollars you spent on N are literally going down the soil drain. Beyond that, nitrogen and phosphorus leaching and runoff from agriculture is responsible for the “dead zone” in the Gulf of Mexico, where those two nutrients feed algae that suck up all the oxygen in the water, killing the other cute ocean creatures. It can also be a major health concern, especially for infants, when high levels of nitrate make it into drinking water. Avoid it!
Volatilization: Ammonium is converted to ammonia gas in hot, windy conditions and/or alkaline soils. Incorporating fertilizers and manures immediately after applying can prevent or reduce losing N to volatilization, so that the N fertilizer you applied actually makes it into your plants.
Understanding the basic N processes will equip you to use and not lose the N fertility that you spend money purchasing and time applying. In addition to getting the best bang for your buck on N fertilizers, your crops will get enough but not too much juice. You can pat yourself on the back for not being an accidental polluter. Win, win, win.
Plants with enough N generally have dark green color on both old and young leaves. When a plant is deficient in N, it can move it from older leaves (making them yellow) into the younger developing ones that need more go-juice. So you will often see N deficiency show up first as yellowing of the older leaves. Crops can also appear stunted (picture tiny little cabbages) or flower (bolt) prematurely. Excess N can look like super dark-green leaves, prolific vegetative growth, delayed or sparse fruiting, weak stems (causing lodging), and even increased disease and pest pressure. Too much of a good thing!
Typically, vegetables use the bulk of their N early in their growth cycle, and they level off on uptake during fruiting. At this stage (allow us to anthropomorphize a bit), some plants perform better under pressure, and a little bit of stress can make them think, “Oh no, I’m running out of N — these halcyon summer days won’t last forever! I’ve got to stop this vegetative lollygagging and look to reproduce.” — Direct quote from Striped German tomato plant #3, Sam’s garden, Minneapolis, MN, August 5, 2025.
Similarly, pruning a cucumber vine can hasten flowering and fruiting, or heavily harvesting mature leaves off of a kale crop can induce fast regeneration of new leaves (the solar panels it needs to start its plant-baby-making). Nothing like a little memento mori as motivation!
Common organic N sources include:
- Blood meal.
- Feather meal.
- Seed meal (alfalfa, soybean).
- Chilean nitrate (beware NOP limits on its use and avoid using in a high tunnel due to high salt content).
- N-fixing bacteria associated with leguminous cover crops.
- Chicken litter (keep in mind, this will come with P, K, Ca…so get an analysis before applying).
- Fish emulsion (note: also comes with other nutrients, check the analysis on the container).
- Compost (caution: not the best way to supply N since it usually means applying too much P. At the very least, get an analysis before applying).
Phosphorus
Whereas N can be very mobile in the soil when it dissolves in water, Phosphorus (P) generally does not move around the soil. Sometimes crops are unable to access P that is plentiful but held tightly to soil particles. Only a small percentage of the P applied through fertilizer is accessible to the crop in that first season. Most of it remains immobilized, bonded to soil minerals until microbes break apart those chemical bonds in later seasons.
When microbes do make the P available to plants, roots can take it up only when they are in contact with the P, since it won’t dissolve in water and flow to the roots. Thus, a biologically active soil and large root systems will render soil P more plant-available. Phosphorus applied through fertilizer will stay mostly where it is laid and might require tillage to move it into a crop’s root zone.
Soil P is less available to plants in acidic (below pH 6) or alkaline (above pH 8) soils. Once in the plant, P is very mobile and can be moved from old to new growth as needed to advance the plant towards reproduction, like N.
P is needed at all stages of growth, especially during flowering and fruiting. Most crops use more than half of their total P after they first flower. We’ll often see P deficiencies in spring, when root systems are small and soils are cold, which slows biological activity. Environmental conditions like soil compaction, pH, waterlogged soil, and others that slow down soil biology and root growth can also hinder the availability of P, even when there is plenty of it in the soil. This is when raised beds, black plastic, and field orientation can assist in improving P availability (insofar as all of these practices influence soil moisture and temperature). P deficiency can make plants look compact, with reddish to purple leaves.
Too much P rarely hurts plants directly, but excessive levels can cause antagonisms with other nutrients, which prevents their absorption into the plant. For example, excess P can reduce potassium uptake (as shown above in photo 1), as well as zinc and iron. Excessive P from fertilizers or compost can also cause salt injury to crops, especially in high tunnels.
Despite the soil’s great ability to hold on to P it can also move. Many of us have heard about the algae blooms in surface waters that are the direct results of fertilizer over-application. Some of that P is obviously not staying put in the soil as we described above. P can be washed into surface waters via soil that is actively leaving the farm (erosion), manure or fertilizer that was applied and left on the soil surface and carried away by rain (runoff), or, if soil P has accumulated beyond the soil’s capacity to hold it, in dissolved water (leaching).
Common organic P sources include:
- Soft rock phosphate (note: finely-ground types, called micronized, are plant-available more quickly).
- Cover crops that absorb soil P will make it available to following cash crops as they break down.
- Manure, pelleted or otherwise.
- Fish emulsion.
- Manure-based compost.
Potassium
Potassium (K) exists in the soil in three forms: unavailable, slowly available, and readily available to the plant. Soil tests measure the readily available K, which is dissolved in the soil solution (soil water) or held on clay particles. As plants take up K from the soil solution, those clay particles release additional K into the soil solution. Increasing soil moisture will therefore increase the plant availability of K, but chronically saturated soils can reduce root activity and K uptake.

Progression of K deficiency symptoms in a high tunnel that had been regularly amended with compost for several years at Michigan State University. The soil tests showed “above optimum” levels of P in the soil. The problem was, despite also having high levels of soil K, leaf tissue sampling indicated that the plant was not taking it up. This was likely caused by high soil P levels interfering with the plant’s ability to absorb soil K. Photo by Sarah Geurkink.
K is, for the most part, held on soil particles and less liable to leach, so farmers can apply it in the spring or fall, before or after the main season. However, K can leach out of soils that have a low Cation Exchange Capacity (CEC). CEC is like your soil’s fuel tank — it determines how many nutrients it can hold. Clayey soils and those with higher organic matter are able hold more nutrients than sandy soils. For sandier, low CEC soils, a K application closer to planting with additional small in-season applications might be a more economical way to amend K. Soil K is less available to plants in acidic soils (below pH 6).
Potassium does all sorts of things for a plant, but in short: crucial for regulating photosynthesis, immunity, structure, drought resistance, respiration, starch protein production, and more. Potassium deficiency can look like stunted plant growth, interveinal chlorosis, reduced yield, and (namely on tomatoes) ripening disorders such as Yellow Shoulder Disorder. These K-deficiency symptoms can be seen when soils have excess P, that makes it hard for the plant to take up K. Excessive K can crowd out calcium and magnesium on soil particles, reducing their availability.
Common organic sources of K include:
- Sulfate of potash (note: also available in water-soluble versions for fertigation, such as Diamond K ultrafines).
- Potassium nitrate (also supplies nitrogen).
- Greensand, aka glauconitic sandstone (also supplies trace minerals).
- Potassium magnesium sulfate, aka Sul-Po-Mag, aka KMag (also contains Mg and S).
- Fish Emulsion (also supplies nitrogen and phosphorus).
To help you remember the basic roles of these 3 primary macronutrients, here is a pithy quip (which is also the name of Sam’s literary tavern) about the primary macros: N grows big green plants equipped with lots of solar panels to power reproduction; P fuels root growth, flowering and fruiting; K bolsters resilience (to cold, heat, drought, disease), crop quality, and storage.
Calcium
Calcium (Ca) is crucial to the plant’s structure. It is a significant component of cell walls, cements cells together, and is needed for actively growing roots and shoots. Ca tends to be present in many native soils, but not all. Because it is not especially mobile in the plant, symptoms of Ca deficiency can appear even when there are adequate levels of it in the soil. Ca is dissolved in water and travels through the plant (through the xylem), as water is drawn from the soil to roots to leaves and then into the atmosphere. This process is called transpiration. So insufficient water, too much water, or rapid plant growth can disrupt Ca supply to specific parts of the plant, especially when those specific plant parts don’t transpire much (fruit). The calcium, which is hitching a ride with the water, is not getting to those parts fast enough. A common example of this is blossom end rot on tomato fruits. Ca is less available to plants in acidic soils (below pH 6).
Common organic sources of Ca include:
- Oyster shells.
- Water soluble calcium (note: fine for fertigation, but it is not super effective to apply Ca via foliar sprays).
- Gypsum (also contains sulfur, but does not significantly affect soil pH).
- Lime (will add calcium to your soil, but only use if you also want to raise your pH).
- Manure, especially egg layer litter (note: contains calcium carbonate, which can steadily raise your pH to a level that is too alkaline for crops).
- Bone meal (also contains N and P).
- Compost (contains many nutrients, get an analysis before applying!).
- Some fish emulsion products.
Sulfur
Sulfur (S) plays a big role in forming proteins and giving flavor to pungent crops like alliums and brassicas. We’ve seen low sulfur on several soil tests recently, and that in part can be attributed to the decrease in industrial air pollution and subsequent rain deposit of the sulfur from the air into the soil (who can complain about that, amiright?). Sulfur is held on organic matter. It is prone to leaching in sandy soils, and is less accessible to plants in acidic soils (below pH 6).
Common organic sources of S include:
- Potassium sulfate (aka Sulfate of potash).
- Potassium magnesium Sulfate (Sul po mag).
- Gypsum (also contains Ca).
- Irrigation water…sometimes (get yours tested).
- Plant residues.
- Leaf litter.
- Manure.
- Compost (contains many nutrients, get an analysis before applying).
- Deep-rooted cover crops can bring up sulfur and prevent its leaching.
Please note: Do not confuse the plant available sulfate with elemental sulfur, which is commonly applied to acidify the soil. Elemental Sulfur is not directly available to plants; it needs to be broken down by soil microbes to become a plant-available sulfate, and this acid reaction will lower your soil pH, which would be an unfortunate surprise if you did not intend it.
Magnesium
Magnesium (Mg) is the main component of chlorophyll, and plays a large role in the plant’s metabolism. Mg is mobile in the plant, and like N and P, the plant can move Mg from older leaves to newly developing ones when it is deficient. Soil Mg is less accessible to plants in acidic soils (below pH 6).
Plants that are Mg deficient are stunted and lose their color. The leaves’ veins might stay green, while the interveinal areas will turn yellow and eventually brown. Because these symptoms can be confused with herbicide drift, disease, or other nutrient deficiencies, it is important to submit a tissue test before amending with Mg. Toxicity symptoms of Mg are stunted roots and shoots, copper-colored leaves, salt injury, and necrotic spots (also the name of Sarah’s punk band).
Common organic sources of Mg include:
- Magnesium sulfate (aka Epsom salts).
- Chelated Mg.
- Sul-po-mag.
- Dolomitic lime (this will also raise your pH, so be sure you want that).
- Irrigation water…sometimes (get yours tested).
Where does compost stand?
Can compost take care of your macronutrient requirements? While it contains some amount of all the macronutrients, compost’s nutrient levels vary widely, even between loads coming from the same supplier. The macronutrients in any one load of compost is probably not in the ratio your soil needs. Additionally, most composts have relatively low levels of nitrogen, and much of that nitrogen is not available to plants in the first year after application.
Should you let that stop you from using compost altogether? Not always — it still has many benefits. But do submit a sample to your local soil lab so you understand what macronutrients you are adding to your soil with compost. Or if you’re buying compost, the manufacturer may be able to provide you with an analysis. Applying lots of compost with an unknown nutrient content, especially to soils already high in nutrients and organic matter, could easily get you in the situation described above (see photo 1): you might inadvertently increase already-high levels of nutrients, causing a series of negative consequences.
We think of compost as an input to improve soil texture, increase soil organic matter (with all the benefits higher SOM includes), and provide longer-term fertility release, rather than a source of in-season fertility.
Conclusion
If you’ve noticed a theme running through the availability of all of the macronutrients, it is that the soil’s biological activity is the star of the show. Microbes transform and release plant-available nutrients. As the soil symphony conductor, your greatest instrument of fertility is to optimize the conditions and shine a spotlight on soil biology. Microbe-positive soil conditions are simple to remember because they are also enjoyed by us humans: temperature, moisture, oxygen, pH, and organic matter.
Readers, thanks for sticking with us. We hope to have convinced you to put a little extra brain space during the off-season into learning about your soil’s nutrient levels and potential so that you can fertilize thoughtfully. At the very least we hope you can now watch out for, identify, and troubleshoot some of the nutrient-related issues you might see next season. Remember that you don’t always have to solve an issue at the height of the season, and keeping records and photos of what you’re seeing can help you figure it out the following winter.
With it being the new year and a time for new beginnings, we’ll share our (completely made up) Golden Rule of Farming: Each year you get one try. This can be scary or liberating – we only get one try, also means we can try again next year. Consider approaching nutrient management with an iterative “continuous improvement” mindset. At the end of the season you can test again to see how things went and tweak the following year’s plan. Try, try again.
Until next time, warm wishes from your soil doulas.
Sarah Geurkink offers production support to commercial organic farms through the Organic Association of Kentucky (OAK). She previously managed organic, diversified farms in Michigan and Washington for 13 years. Sarah especially enjoys learning from farmers, sharing ideas, and solving production challenges. You can reach her at sarah@oak-ky.org.
Sam Oschwald Tilton is a long-time veg head and an organic agronomist. He helps new and established farmers improve their weed management, soil fertility, and machinery. He also works with organizations to deliver hands-on trainings and field days. Sam lives in Minneapolis and enjoys gardening with the neighborhood. He is happy to hear from you, at glacialdrift@protonmail.com.
Copyright Growing For Market Magazine.
All rights reserved. No portion of this article may be copied
in any manner for use other than by the subscriber without
permission from the publisher.











